Piggott, Luke, Silva, Ana  ORCID: https://orcid.org/0000-0002-1184-4909, Robinson, Timothy, Santiago-Gomez, Angelica, Simoes, Bruno M., Becker, Michael, Fichtner, Iduna, Andera, Ladislav, Young, Philippa, Morris, Christine, Barret-Lee, Peter, Alchami, Fouad, Piva, Marco, Vivanco, Maria d. M., Clarke, Robert B., Gee, Julia ORCID: https://orcid.org/0000-0002-1184-4909, Robinson, Timothy, Santiago-Gomez, Angelica, Simoes, Bruno M., Becker, Michael, Fichtner, Iduna, Andera, Ladislav, Young, Philippa, Morris, Christine, Barret-Lee, Peter, Alchami, Fouad, Piva, Marco, Vivanco, Maria d. M., Clarke, Robert B., Gee, Julia  ORCID: https://orcid.org/0000-0001-6483-2015 and Clarkson, Richard ORCID: https://orcid.org/0000-0001-6483-2015 and Clarkson, Richard  ORCID: https://orcid.org/0000-0001-7389-8673
2018.
Acquired resistance of ER-positive breast cancer to endocrine treatment confers an adaptive sensitivity to TRAIL through post-translational downregulation of c-FLIP.
Clinical Cancer Research
24
(10)
, pp. 2452-2463.
10.1158/1078-0432.CCR-17-1381 ORCID: https://orcid.org/0000-0001-7389-8673
2018.
Acquired resistance of ER-positive breast cancer to endocrine treatment confers an adaptive sensitivity to TRAIL through post-translational downregulation of c-FLIP.
Clinical Cancer Research
24
(10)
, pp. 2452-2463.
10.1158/1078-0432.CCR-17-1381

|
Preview |
PDF
- Accepted Post-Print Version
Download (363kB) | Preview |
![Figure 1 [thumbnail of Figure 1]](https://orca.cardiff.ac.uk/108076/7.hassmallThumbnailVersion/Figure-1%20Final.png)  Preview |
Image (PNG) (Figure 1)
- Supplemental Material
Download (87kB) | Preview |
![Figure 2 [thumbnail of Figure 2]](https://orca.cardiff.ac.uk/108076/8.hassmallThumbnailVersion/Figure-2%20Final.png) 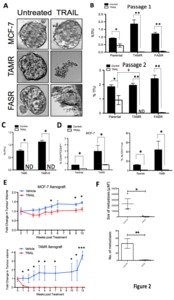 Preview |
Image (PNG) (Figure 2)
- Supplemental Material
Download (2MB) | Preview |
![Figure 3 [thumbnail of Figure 3]](https://orca.cardiff.ac.uk/108076/9.hassmallThumbnailVersion/Figure-3%20Final.png) 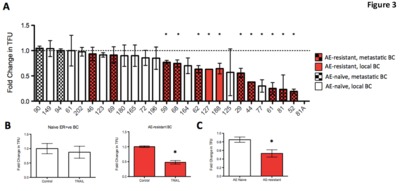 Preview |
Image (PNG) (Figure 3)
- Supplemental Material
Download (527kB) | Preview |
![Figure 4 [thumbnail of Figure 4]](https://orca.cardiff.ac.uk/108076/10.hassmallThumbnailVersion/Figure-4%20Final.png)  Preview |
Image (PNG) (Figure 4)
- Supplemental Material
Download (932kB) | Preview |
![Figure 5 [thumbnail of Figure 5]](https://orca.cardiff.ac.uk/108076/11.hassmallThumbnailVersion/Figure-5%20Final.png) 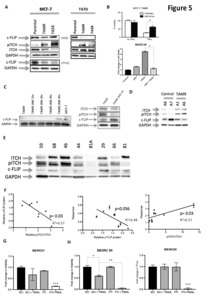 Preview |
Image (PNG) (Figure 5)
- Updated Version
Download (1MB) | Preview |
Preview |
PDF (Figure S1)
- Supplemental Material
Download (863kB) | Preview |
Abstract
Purpose One-third of ER-positive breast cancer patients who initially respond to endocrine therapy become resistant to treatment. Such treatment failure is associated with poor prognosis and remains an area of unmet clinical need. Here we identify a specific post-translational modification that occurs during endocrine resistance and which results in tumour susceptibility to the apoptosis inducer TNF-Related Apoptosis-Inducing Ligand (TRAIL). This potentially offers a novel stratified approach to targeting endocrine resistant breast cancer. Experimental Design Cell line and primary-derived xenograft models of endocrine resistance were investigated for susceptibility to TRAIL. Tumour viability, cancer stem cell (CSC) viability (tumourspheres), tumour growth kinetics and metastatic burden were assessed. Western blots for the TRAILpathway inhibitor, c-FLIP, and upstream regulators were performed. Results were confirmed in primary culture of 26 endocrine-resistant and endocrine-naïve breast tumours. Results Results Breast cancer cell lines with acquired resistance to tamoxifen-(TAMR) or faslodex were more sensitive to TRAIL than their endocrine-sensitive controls. Moreover, TRAIL eliminated CSClike activity in TAMR cells, resulting in prolonged remission of xenografts in vivo. In primary culture, TRAIL significantly depleted CSCs in 85% endocrine-resistant, compared with 8% endocrine-naïve tumours, while systemic administration of TRAIL in endocrine-resistant patient-derived xenografts reduced tumour growth, CSC-like activity and metastases. Acquired TRAIL sensitivity correlated with a reduction in intra-cellular levels of c-FLIP, and an increase in Jnk-mediated phosphorylation of E3-ligase, ITCH, which degrades c-FLIP. Conclusions These results identify a novel mechanism of acquired vulnerability to an extrinsic cell death stimulus, in endocrine resistant breast cancers, which has both therapeutic and prognostic potential.
| Item Type: | Article |
|---|---|
| Date Type: | Publication |
| Status: | Published |
| Schools: | Schools > Pharmacy Schools > Medicine Schools > Biosciences Research Institutes & Centres > European Cancer Stem Cell Research Institute (ECSCRI) |
| Publisher: | American Association for Cancer Research |
| ISSN: | 1078-0432 |
| Date of First Compliant Deposit: | 10 January 2018 |
| Date of Acceptance: | 16 January 2018 |
| Last Modified: | 06 Nov 2024 14:16 |
| URI: | https://orca.cardiff.ac.uk/id/eprint/108076 |
Citation Data
Cited 23 times in Scopus. View in Scopus. Powered By Scopus® Data
Actions (repository staff only)
 |
Edit Item |




 Altmetric
Altmetric Altmetric
Altmetric